About the FDA Adverse Event Database: Real-World Insights
The FDA in America has an adverse event database that tracks drug-related events. Drug-to-drug interactions occur when two or more medications interact in ways that can alter their effectiveness or increase the risk of adverse reactions, potentially leading to serious outcomes such as hospitalisation, disability, or life-threatening events.
Methodology: Using DuckDB, SQL and Python for Data Analysis
Firstly, It was a technical challenge to run queries on nearly 500 GB of data on a 2TB Mac laptop already filled with LLM downloads. The complete dataset is 460GB across 1,572 partitioned files. Due to its size, it wouldn’t fit efficiently in a PostgreSQL database. Instead, we used DuckDB to handle querying, along with extensive SQL and some Python to process the data. These tools allow us to extract and analyze the information without compromising performance.
The technical details, including Python scripts and SQL queries used, are yet to be documented in a followup post on our GitHub repository. DuckDB has been essential for managing the complexity and scale of the dataset.
Data Cleaning: Removing Anomalous Records for Accurate Drug Safety Insights
While analysing the data set, we found some events that included over 180 drugs in a single report—likely erroneous data. We made the assumption to filter those out. We also precomputed certain columns rather than relying solely on nested JSON queries to speed up analytics. The patientonsetage has a few extreme values as well therefore we set the range on the ages between 0 and 120. We didn’t use this method throughout and some of the data you will see is erroneous such as the years when the event was reported.
Cases by year
The number of adverse drug event reports has significantly increased over time, with recent years showing the highest volumes. In 2021, the data records approximately 1.7 million cases, making it the peak year in the dataset. Similarly, 2020, 2019, and 2018 each saw over 1.4 million cases. The upward trend may reflect various factors, including improved reporting systems, increased drug use, or regulatory changes. However, there is a noticeable drop in cases after 2021, with the data for 2023 and 2024 showing fewer reports. Earlier years, particularly before 2003, have much lower counts, likely due to underreporting or limited data availability from that period. The data also includes isolated cases with future or erroneous years, which may require further investigation.
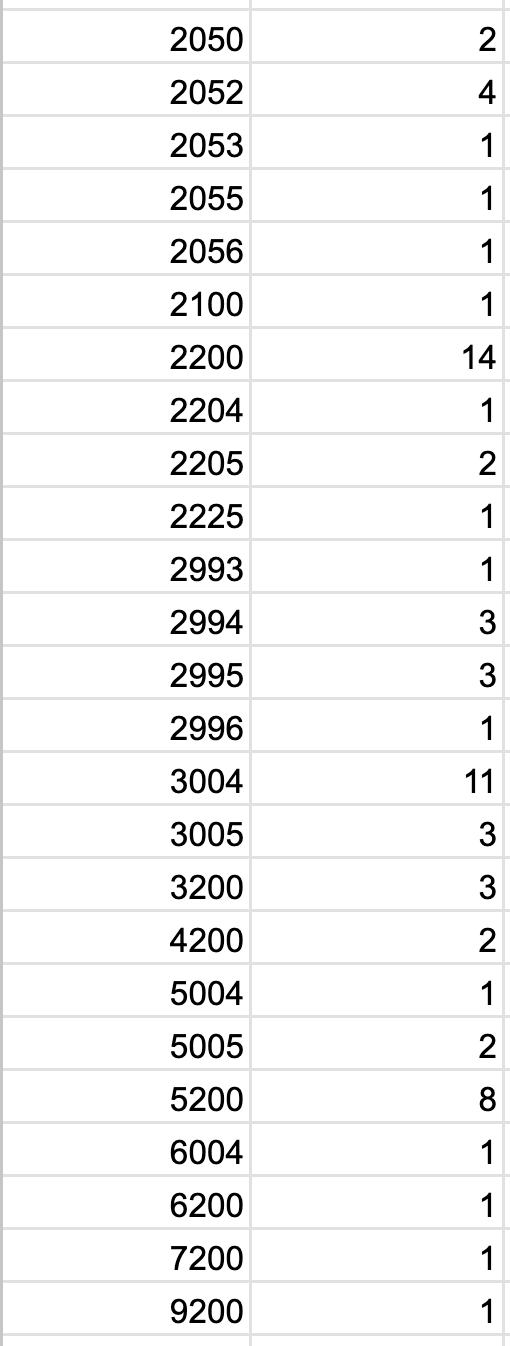
Here you can see some of the dates that didn’t not make sense.
Key Patient Demographics: Age, Gender, and Risk Factors
The adverse drug event data includes detailed information about patient demographics, highlighting variations in hospitalization, death, disabling events, and life-threatening outcomes across gender and age groups. Older patients, particularly those over 61, show the highest percentages of death events, with males aged 81+ reporting a 23.18% death rate and females in the same age group showing a 16.96% rate. Hospitalization percentages also increase with age, reaching over 34% for both genders in the 81+ group.
For younger patients, the percentages vary widely. Infants (0-1 years) exhibit high hospitalization rates of over 30% across genders but relatively lower percentages for death events compared to older populations. Reports with unknown gender show similar patterns, though the percentage of events like congenital anomalies is noticeably higher for certain age groups, particularly those under one year.
The data points to clear demographic trends in reported events, although further analysis would be required to understand the underlying causes. These breakdowns provide insights into how age and gender may relate to the types of reported adverse outcomes.
We didn’t include the unknowns here as made the chart quite difficult visually
Top Administration Routes: Oral, Intravenous, and More
The data on administration routes reveals significant variation in the frequency and severity of reported adverse events. The oral route, with over 32 million total events, accounts for a large proportion of cases, including a 32.55% hospitalization rate and an 11.43% death rate. Intravenous administration has the second-highest number of events and a higher percentage of serious outcomes, with a 37.21% hospitalization rate and a 14.5% death rate. Intramuscular and rectal routes also display relatively high rates of hospitalization, at 35.75% and 41.33% respectively.
Routes such as subcutaneous and topical show lower event volumes and severity percentages, while rarer routes like intravitreal and intra-arterial demonstrate elevated rates of death and life-threatening events despite lower total case numbers. Notably, transmucosal administration, though reporting fewer events overall, has the highest death percentage at 56.95%.
These data patterns provide insight into how different administration routes are associated with varying risks of adverse events, without drawing any clinical conclusions. Further analysis may help explain the trends observed in the data
Polypharmacy Insights: How Multiple Drugs Affect Patient Safety
The FDA’s adverse event data shows a trend where the reported likelihood of death increases as the number of medications taken rises. This trend is visible when analyzing cases with different numbers of drugs involved. The data presents a pattern that may provide further insight into how multiple medications are associated with serious outcomes.
We have drawn a trend line based chart around females and events ending in death. The trend line illustrates this correlation, highlighting the increased occurrence of reported deaths as the number of medications grows.
Drug Class Statistics: High-Risk Medications and Their Impact
Our analysis of the FDA’s adverse event database involves running aggregate SQL queries to examine the distribution of adverse drug reactions (ADRs) across different drug classes. Certain classes, including anticoagulants and opioids, frequently appear in reports associated with serious outcomes such as internal bleeding and respiratory issues. These findings are based on the frequency and patterns observed in the data, without further interpretation. The dataset serves as a resource for identifying which drug classes are most commonly reported in connection with adverse events.
Key Congenital and Serious Adverse Reactions in FDA Drug Reports
The FDA’s adverse event data reveals patterns in both congenital and serious reactions reported by patients and healthcare providers. Congenital reactions primarily involve drug exposure during pregnancy, leading to conditions such as congenital anomalies, birth defects, and premature births. Serious reactions are often linked to drug ineffectiveness, death, and severe complications like pneumonia and dyspnoea. This section provides an overview of the most frequently reported reactions, with data presented to highlight trends rather than draw medical conclusions.
Reports frequently cite “Foetal exposure during pregnancy,” congenital anomalies, and developmental delays among the most common reactions.
Congenital Reaction Data:
Reports frequently cite “Foetal exposure during pregnancy,” congenital anomalies, and developmental delays among the most common reactions.
Serious Reaction Data:
The most reported serious reactions include “Drug ineffective,” “Death,” and severe outcomes such as pneumonia and falls, with high percentages of serious classifications.
Common Drug Combinations Linked to Fatal Events
Drug interactions play a crucial role in patient safety, particularly when multiple high-risk medications are prescribed together. Our analysis of the FDA’s adverse event database highlights the most frequently reported four-drug combinations in cases flagged as deaths. Among the most common groupings are:
- Cetirizine, Leflunomide, Methotrexate, and Rituximab (1,344 occurrences)
- Cyclophosphamide, Doxorubicin, Prednisone, and Rituximab (1,185 occurrences)
- Bortezomib, Cyclophosphamide, Dexamethasone, and Melphalan (952 occurrences)
- Ampicillin, Ceftriaxone, Piperacillin/Tazobactam, and Vancomycin (720 occurrences)
While these findings do not establish causation, they highlight frequent multi-drug interactions associated with severe outcomes. As the number of drugs in a regimen increases, so does the complexity of potential interactions. These insights provide a foundation for further investigation into the risks of specific drug combinations, helping to inform clinicians and researchers focused on medication safety.
Conclusion
- Rising ADR Reports – The number of reported adverse drug reactions (ADRs) has been steadily increasing, highlighting a growing concern for patient safety.
- Elderly at Higher Risk – Patients over 61 are disproportionately affected, with higher rates of hospitalisation and fatal outcomes.
- Administration Matters – Drugs given intravenously (IV) are linked to more severe reactions compared to oral medications.
- Need for Vigilance – Healthcare providers must prioritize careful prescribing, especially for older adults and high-risk medications.
- Enhanced Patient Education – Informing patients about potential drug side effects can help prevent severe reactions.
- Stronger Monitoring & Reporting – Improving adverse event tracking and encouraging timely reporting can save lives.
- Next Steps – The healthcare industry must push for better drug safety measures, updated prescribing guidelines, and AI-driven solutions to detect ADR patterns earlier.
Bottom Line: ADRs are a silent threat, but with proactive strategies, we can significantly reduce harm and improve patient outcomes.
Interested in safer prescribing for older patients?
We’re exploring how technology can support medication reviews, reduce adverse drug interactions, and improve prescribing decisions using approaches like STOPP/START criteria.
If you’re involved in geriatric prescribing, medication safety, or clinical decision support, we’d love to hear your thoughts.
Get in touch: Contact us to discuss AI-driven medication safety and prescribing support.

